Unlocking the Intricacies: The Role of KNDy Neurons Expressing Kisspeptin, Neurokinin B, and Dynorphin in Reproductive Physiology
The realm of reproductive physiology is a complex interplay of intricate signaling pathways, where a delicate balance is maintained to ensure the proper functioning of the reproductive system. Among the various players in this symphony, KNDy neurons, expressing the trifecta of Kisspeptin, Neurokinin B (NKB), and Dynorphin, have emerged as key orchestrators. This article delves into the multifaceted roles of KNDy neurons and their crucial contributions to the regulation of reproductive processes.
Understanding KNDy Neurons:
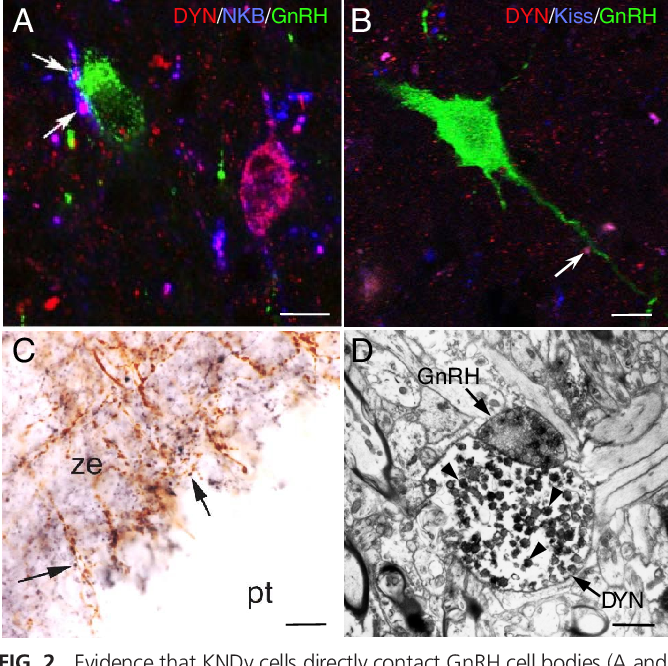
KNDy neurons are a subset of neurons found in the arcuate nucleus of the hypothalamus, a region of the brain known for its pivotal role in regulating hormonal balance. The acronym KNDy stands for the co-expression of three neuropeptides: Kisspeptin, Neurokinin B, and Dynorphin. These neuropeptides act in concert to modulate the secretion of gonadotropin-releasing hormone (GnRH), a master regulator of the reproductive axis.
Kisspeptin: The Ignition Switch
Kisspeptin, encoded by the KISS1 gene, is a potent stimulator of GnRH release. It acts as the ignition switch for the reproductive system, playing a central role in initiating puberty and regulating the menstrual cycle. Kisspeptin binds to its receptor, G protein-coupled receptor 54 (GPR54), triggering a cascade of events that culminate in the release of GnRH. This process is pivotal for the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), essential for ovulation and spermatogenesis.
Neurokinin B: The Synchronization Signal
Neurokinin B, encoded by the TAC3 gene, is another critical player in the orchestration of reproductive events. This neuropeptide acts in synergy with Kisspeptin, contributing to the pulsatile release of GnRH. The synchronized action of Neurokinin B and Kisspeptin is essential for the proper regulation of the menstrual cycle and the maintenance of reproductive health. Dysregulation of Neurokinin B signaling has been implicated in conditions such as hypothalamic amenorrhea and polycystic ovary syndrome (PCOS).
Dynorphin: The Brakes on Reproduction
In contrast to the stimulatory effects of Kisspeptin and Neurokinin B, Dynorphin, encoded by the PDYN gene, acts as a brake on the reproductive axis. Dynorphin is an opioid neuropeptide that binds to kappa opioid receptors (KORs) on GnRH neurons, inhibiting their activity. This inhibitory effect is crucial for preventing excessive GnRH release and maintaining the delicate balance between the stimulatory and inhibitory signals within the reproductive system.
Role of KNDy Neurons in Puberty:
Puberty marks the transition from childhood to reproductive maturity and is tightly regulated by the interplay of various hormonal signals. KNDy neurons play a central role in the initiation of puberty through the release of Kisspeptin. The pulsatile secretion of Kisspeptin stimulates the release of GnRH, which, in turn, triggers the secretion of gonadotropins and the development of secondary sexual characteristics.
Disruptions in the function of KNDy neurons or their associated neuropeptides can lead to disorders of puberty, such as precocious puberty or delayed puberty. Understanding the nuances of KNDy neuron function provides insights into the underlying mechanisms of these conditions and opens avenues for potential therapeutic interventions.
Menstrual Cycle Regulation:
The menstrual cycle is a complex interplay of hormonal events orchestrated by the hypothalamus, pituitary gland, and ovaries. KNDy neurons, through the coordinated action of Kisspeptin and Neurokinin B, regulate the pulsatile release of GnRH, which, in turn, governs the secretion of LH and FSH. This intricate dance of hormones ensures the proper development and release of mature ova from the ovaries, setting the stage for fertility.
Dysregulation of KNDy neuron function can lead to menstrual irregularities, including anovulation and amenorrhea. These disruptions have implications for female reproductive health, impacting fertility and overall well-being. Researchers are exploring the therapeutic potential of modulating KNDy neuron activity to address reproductive disorders and improve outcomes for individuals facing challenges in conception.
Clinical Implications and Therapeutic Potential:
The pivotal role of KNDy neurons in reproductive physiology has sparked interest in targeting this system for therapeutic interventions. Understanding the delicate balance between stimulatory (Kisspeptin and Neurokinin B) and inhibitory (Dynorphin) signals allows researchers to explore novel approaches for managing conditions such as infertility, polycystic ovary syndrome (PCOS), and hypothalamic amenorrhea.
Selective modulation of KNDy neuron activity holds promise for the development of targeted therapies that can restore hormonal balance in individuals with reproductive disorders. Pharmacological agents that enhance Kisspeptin or Neurokinin B signaling, or inhibit Dynorphin action, could offer new avenues for managing conditions associated with impaired fertility.
Conclusion:
In the intricate tapestry of reproductive physiology, KNDy neurons expressing Kisspeptin, Neurokinin B, and Dynorphin stand out as crucial conductors. Their harmonious interplay regulates the pulsatile release of GnRH, orchestrating the symphony of hormonal events that drive puberty, menstrual cycle regulation, and fertility. As researchers delve deeper into the complexities of KNDy neuron function, the potential for unlocking novel therapeutic interventions to address reproductive disorders becomes increasingly promising. The journey to unravel the mysteries of KNDy neurons continues, holding the key to improved reproductive health for individuals worldwide.
-
What are KNDy neurons, and where are they located?
- KNDy neurons are a subset of neurons located in the arcuate nucleus of the hypothalamus, a region in the brain responsible for regulating hormonal balance.
-
What is the significance of Kisspeptin in reproductive physiology?
- Kisspeptin is a neuropeptide that acts as a potent stimulator of gonadotropin-releasing hormone (GnRH) release. It plays a central role in initiating puberty and regulating the menstrual cycle.
-
How does Neurokinin B contribute to reproductive processes?
- Neurokinin B acts in synergy with Kisspeptin to modulate the pulsatile release of GnRH. This synchronization is crucial for the regulation of the menstrual cycle and the maintenance of reproductive health.
-
What is the role of Dynorphin in the context of KNDy neurons?
- Dynorphin, an opioid neuropeptide, acts as an inhibitory signal. It binds to kappa opioid receptors on GnRH neurons, acting as a brake on reproductive processes and preventing excessive GnRH release.
-
How do KNDy neurons contribute to the initiation of puberty?
- KNDy neurons, through the release of Kisspeptin, play a central role in initiating puberty. The pulsatile secretion of Kisspeptin stimulates the release of GnRH, triggering the hormonal events associated with puberty.
-
What happens when there is a disruption in KNDy neuron function?
- Disruptions in KNDy neuron function can lead to disorders of puberty, such as precocious puberty or delayed puberty. Menstrual irregularities, including anovulation and amenorrhea, can also result from dysregulation of these neurons.
-
How does the menstrual cycle regulation involve KNDy neurons?
- KNDy neurons regulate the pulsatile release of GnRH, which, in turn, governs the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). This orchestration ensures the proper development and release of mature ova from the ovaries.
-
What are the clinical implications of KNDy neuron research?
- Understanding the role of KNDy neurons has implications for managing conditions such as infertility, polycystic ovary syndrome (PCOS), and hypothalamic amenorrhea. Targeting KNDy neuron activity could lead to novel therapeutic interventions.
-
Are there potential treatments targeting KNDy neurons for reproductive disorders?
- Researchers are exploring pharmacological agents that selectively modulate KNDy neuron activity. These approaches include enhancing Kisspeptin or Neurokinin B signaling and inhibiting Dynorphin action, with the aim of developing targeted therapies for reproductive disorders.
-
How might understanding KNDy neurons contribute to improved reproductive health outcomes?
- Unraveling the complexities of KNDy neuron function holds the promise of developing targeted therapies that can restore hormonal balance in individuals with reproductive disorders. This knowledge could contribute to improved fertility and overall reproductive health.






